A Rise in Botanical Supplements
Walking into a supermarket today or shopping online, it is hard to miss the shift in the types of products for sale such as matcha coffees, lion’s mane teas, ginseng soft drinks and ashwagandha gummies. All these products stem from the meteoric rise of functionalism in our foods and beverages. And whilst these products are often supercharged by social media trends, it is more embedded in a population that is seeking to be healthier through what they eat and drink.
Botanicals are part of that uptake. Previously associated as alternative ancient medicine but now continuously sought out by a population seeking to improve their immune health, sleep, cognitive function, and overall wellbeing 1. There is no internationally shared definition for the term ‘botanical’; however, according to the European Food Safety Authority (EFSA) a botanical can be seen as herbs, roots, flowers, mushrooms and other plant-derived extracts 2. Other jurisdictions classify these ingredients differently – such as dietary supplements in the US, functional foods or Kampo medicines in Japan, or traditional herbal products in India and China – reflecting the lack of global consensus on how botanicals are categorised 3.
Despite this regulatory heterogeneity, the economic significance of botanicals is substantial. The US saw its botanical supplement industry reach USD 13.57 billion in 2024 4, while the European herbal market was valued at approximately USD 7.5 billion in 2023 5. Latin America’s herbal supplement market stood at USD 3.39 billion in 2024 6, and Asia-Pacific represents the fastest-growing region globally, driven by deep-rooted traditions in herbal medicine across China, India, and Japan 7. As the approach to ageing shifts from living longer to living healthier for longer, botanicals will likely continue to soar in popularity. This growing demand for botanical dietary supplements stems from positive attitudes toward ‘natural’ products and an expanding body of evidence supporting their beneficial effects on human health.
Plants are rich sources of diverse bioactive compounds, including coumarins, flavonoids, phenolics, alkaloids, terpenoids, tannins, essential oils, lectins, polypeptides, and polyacetylenes; and consequently, their potential health benefits are unsurprising 8. Ashwagandha (Withania somnifera) has been shown to reduce perceived stress and anxiety in adults 9, giving clinical weight to its reputation as an adaptogen. Turmeric (Curcuma longa), a curcuminoid-rich plant, has meaningful reductions in pain and improvements in physical function reported among people with knee osteoarthritis 10. Elderberry (Sambucus nigra), rich in flavanols, has been found to reduce duration and severity of common cold and flu symptoms 11.
Despite these positives, caution is warranted when botanicals are used for medicinal purposes. Their “natural” origin often leads consumers to perceive them as inherently benign, fostering assumptions of safety that are not always justified. This perception has contributed to widespread misunderstanding surrounding botanical products and their appropriate use.

A Credibility Issue
Consider Emily. Like many Americans, she has begun to struggle with knee pain linked to chronic inflammation. On the advice of friends and a growing body of academic research, she adds turmeric to her routine as part of a broader, holistic approach. The label states a meaningful dose of curcuminoids, the “active ingredient” most often linked to turmeric’s health benefits. Weeks later, little has changed, and independent testing reveals that the product contains far less curcuminoids than advertised. Emily does not blame processing choices or the environmental conditions in which the turmeric was grown. She blames the plant itself, stops trusting botanical supplements altogether and possibly takes to social media where it can amplify distrust far beyond a single purchase.
Emily’s story, while hypothetical, reflects a real and measurable problem. Large-scale global assessments of nearly 6,000 commercial herbal products across 37 countries have found roughly 27% were adulterated in some way, illustrating how unmanaged variability and poor verification can erode confidence across the category 12. The answers to seemingly simple questions, what exactly is in the product, how consistent it is batch to batch and whether it reflects what the label claims are not always clear and the solutions are more complex.
Plants are characteristically intricate organisms, and their biological makeup is inherently linked to the abiotic and biotic factors it experiences during growth 13. For example, anyone who has tasted oranges grown in different parts of the world will recognise the effect of geography. Fruits from warmer, sunnier regions often taste sweeter, while those grown in cooler environments can be sharper or more acidic – despite all being, unequivocally oranges. Soil composition, rainfall, temperature, and harvest timing subtly but consistently shapes flavour.
The same principle applies to botanical supplements; species such as ashwagandha or ginseng, which are cultivated across multiple geographic regions, can differ meaningfully in their chemical profiles depending on where and how they are grown. Lan et al.14 showed that Panax ginseng cultivated under different forest canopies, coniferous, broad-leaved, and mixed forests exhibited statistically significant differences in both total ginsenoside content and individual ginsenosides. These subtle changes matter when products are expected to deliver certain health outcomes.
The challenge is compounded by the way botanical products are processed. Raw materials are often sourced from multiple growers and processed in different ways, even within the same supply chain. For example, Kumar et al.15 in Withania somnifera (Ashwagandha) the principal bioactive compounds such as withaferin A, withanones, and withanosides are unevenly distributed across plant parts, with leaves enriched in certain withanolides and roots in withanosides. This means products made from roots, leaves, or whole plants can contain markedly different profiles of these compounds. As a result, two products derived from the same plant but processed differently can vary meaningfully in both composition, potential benefit and therefore quality.
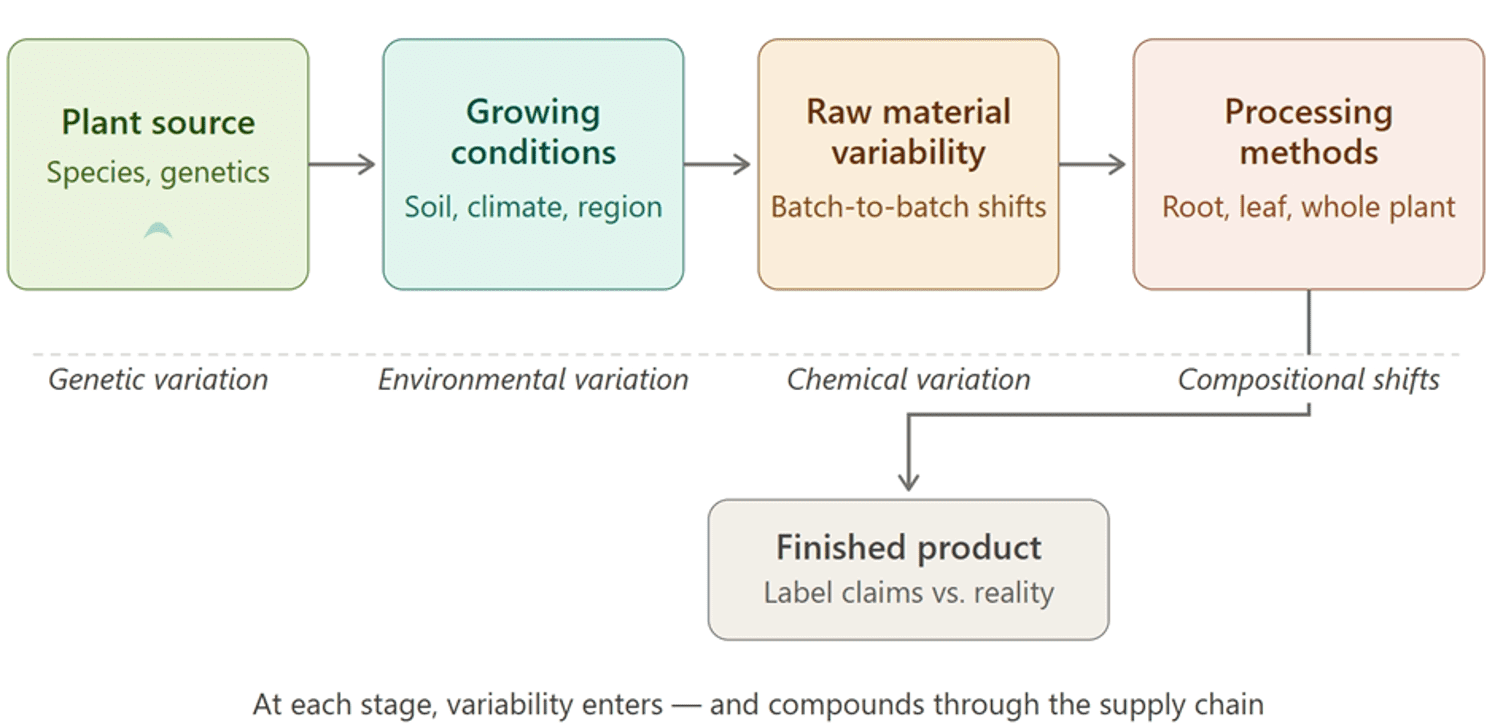
Figure 1 – From plant to product: where botanical variability enters the supply chain. At each stage from genetics, through growing conditions and sourcing to processing – new sources of variability are introduced and compound through to the finished product.
When identity and composition are not carefully verified, variability can slide into misidentification or even deliberate substitution. Cheaper materials may be used to imitate more valuable ingredients, such as peanut skins being substituted for the A-type proanthocyanidins associated with cranberries 16. These practices do more than mislead consumers: they risk undermining efficacy claims and, in some cases, introduce safety concerns that neither the label nor the user expects. Managing these risks calls for a different way of thinking about quality. Modern analytical science offers the tools to make that complexity manageable.
Overview of Analytical Techniques
For much of history, botanical quality was judged by eye and by a small number of simple tests. A root or leaf was inspected to see if it “looked right,” and one or two marker compounds were measured to suggest identity or strength. These methods are still useful and, in many settings, perfectly adequate.
However, modern botanical supply chains are far more complex. Ingredients are grown in different regions, across seasons, and under subtly different environmental and processing conditions. In this context, analytical techniques play a critical role in reducing uncertainty about raw materials, verifying authenticity of source ingredients, and supporting regulatory compliance across different markets. These techniques increasingly fall into two broad categories: 1. targeted methods, which measure predefined metabolites, and 2. untargeted methods, which characterise the overall bio-chemical system.
Targeted Analytical Techniques
Targeted approaches are designed to answer focused questions: Is compound X present? Is it within specification? They rely on prior knowledge of marker compounds, specific chemical constituents used to verify the identity, purity, or potency of a botanical material, as well as reference standards. As such, they are central to regulatory compliance and routine quality control.
Gas Chromatography–Mass Spectrometry (GC–MS)
GC–MS examines the volatile fraction of a botanical. The sample is vapourised and carried by an inert gas through a narrow column, where compounds separate based on their interaction with the stationary phase. As each compound exits the column, it is ionised and fragmented, producing a characteristic mass spectrum 17.
This combination of separation and structural identification makes GC-MS ideal for aroma profiling, characterisation of , residual solvents, and small volatile adulterants. Its limitation is fundamental: larger bioactive phytochemicals (BAPs), including glycosides, polyphenols, curcuminoids, ginsenosides, and withanolides are non-volatile and thermally unstable. This means they fall outside the analytical scope of GC-MS unless chemically derivatised 17.
High-Performance Liquid Chromatography (HPLC)
HPLC uses high pressure or ultra-high pressure (UPLC) to push a liquid sample through a packed column, separating compounds based on their polarity due to the interaction between the stationary and mobile phases. Detection is most often optical (e.g., UV/Visible), and methods are engineered around compounds with known chromatographic behaviour 18. Its strengths are precision, reproducibility, and scalability across laboratories (Figure 2). By design, however, it only detects chemistry the method defines; compounds outside that analytical window remain undetected.
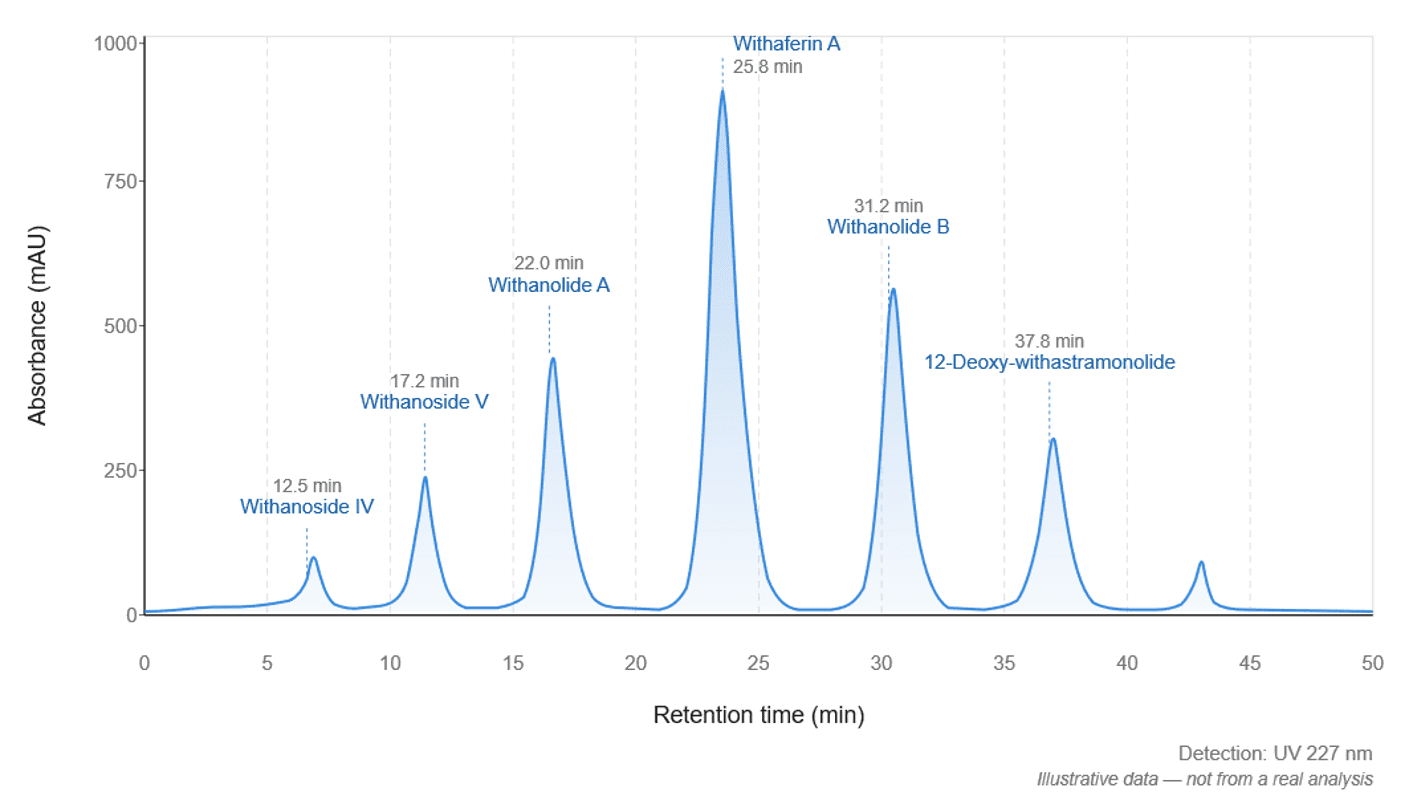
Figure 2 – Illustrative HPLC-UV chromatogram of an ashwagandha (Withania somnifera) root extract at 227 nm, showing separation of six withanolides marker compounds. Retention times and peak heights are simulated for illustrative purposes and do not represent real analytical data.
Liquid Chromatography Mass Spectrometry (LC–MS)
When liquid chromatography is coupled to mass spectrometry, chromatographic peaks gain molecular specificity. Features are defined not only by retention time but by their mass-to-charge ratio and fragmentation pattern. When a photodiode array (PDA) detector is also coupled, UV/visible absorbance data can provide additional confirmation. In practice, this configuration is most applied as a targeted technique used to confirm and, depending on the type of mass analyser, quantify predefined analytes 18.
Untargeted Analytical Techniques
Untargeted techniques shift the analytical question. Rather than confirming the presence of specific compounds, they aim to capture the overall chemical composition of a botanical.
High-Resolution (LC–MS)
When liquid chromatography is paired with high-resolution mass analysers — such as Time-of-Flight (ToF), Orbitrap, or hybrid Q-ToF instruments — the analytical window opens significantly. These platforms measure mass with enough accuracy to infer molecular formulas directly from the data 19.
Depending on how they are configured, high-resolution LC/MS systems can operate in both targeted and untargeted modes: confirming known compounds or scanning broadly for hundreds to thousands of features without preselecting what to look for 20. This flexibility makes them especially valuable in botanical quality assessment, where the question is not just “is compound X present?” but “does this material look like the real thing?”. The resulting datasets are complex, but they become meaningful when compared against well-characterised reference profiles of genuine plant material.
NMR (Nuclear Magnetic Resonance) Spectroscopy
NMR places a prepared extract – whose composition reflects the extraction protocol used – in a strong magnetic field and probes how atomic nuclei respond to radiofrequency energy. Each chemical functional group produces a distinct resonance, and the resulting spectrum integrates signals from all major molecular populations at once 21. Because signal intensity is directly proportional to concentration, NMR is inherently quantitative and highly reproducible for major metabolites above its detection threshold, though sensitivity limits its ability to quantify trace-level components. The result is a holistic chemical portrait that shifts with any meaningful change in composition, making it powerful for assessing overall integrity.
Modern analytical techniques make it possible to measure botanical chemistry in detail. Yet, when used in isolation, each still answers only a narrow question. In practice, no single method can fully describe a living system shaped by species, soil, climate, and processing. What has emerged in response is metabolomics: an approach that treats botanical ingredients not as collections of isolated compounds, but as integrated bio-chemical systems.
Metabolomics: Beyond Markers to Fingerprints
Metabolomics describes the comprehensive measurement of all small molecules present in a biological system at a given moment. In plants, this means capturing the full spectrum of sugars, organic acids, amino acids, phenolics, terpenoids, and countless other metabolites that collectively define a species and reflect how it has grown, been harvested, and processed 18.
Unlike traditional quality control, which reduces a complex extract to the concentration of one or two “marker” compounds, metabolomics treats each botanical as a multidimensional chemical fingerprint. Using an untargeted approach, thousands of features are measured simultaneously.
This shift also changes the underlying question being asked. Rather than asking whether a predefined compound falls within an acceptable range, metabolomic approaches ask whether the overall chemical behaviour of a material is consistent with that of an authentic plant. In a conventional single‑marker framework, an extract is evaluated against one target analyte — for example, whether compound X meets specification. As illustrated in Figure 3a, an authentic botanical extract and a deliberately spiked product can both pass this test, even though their underlying chemistry is fundamentally different.
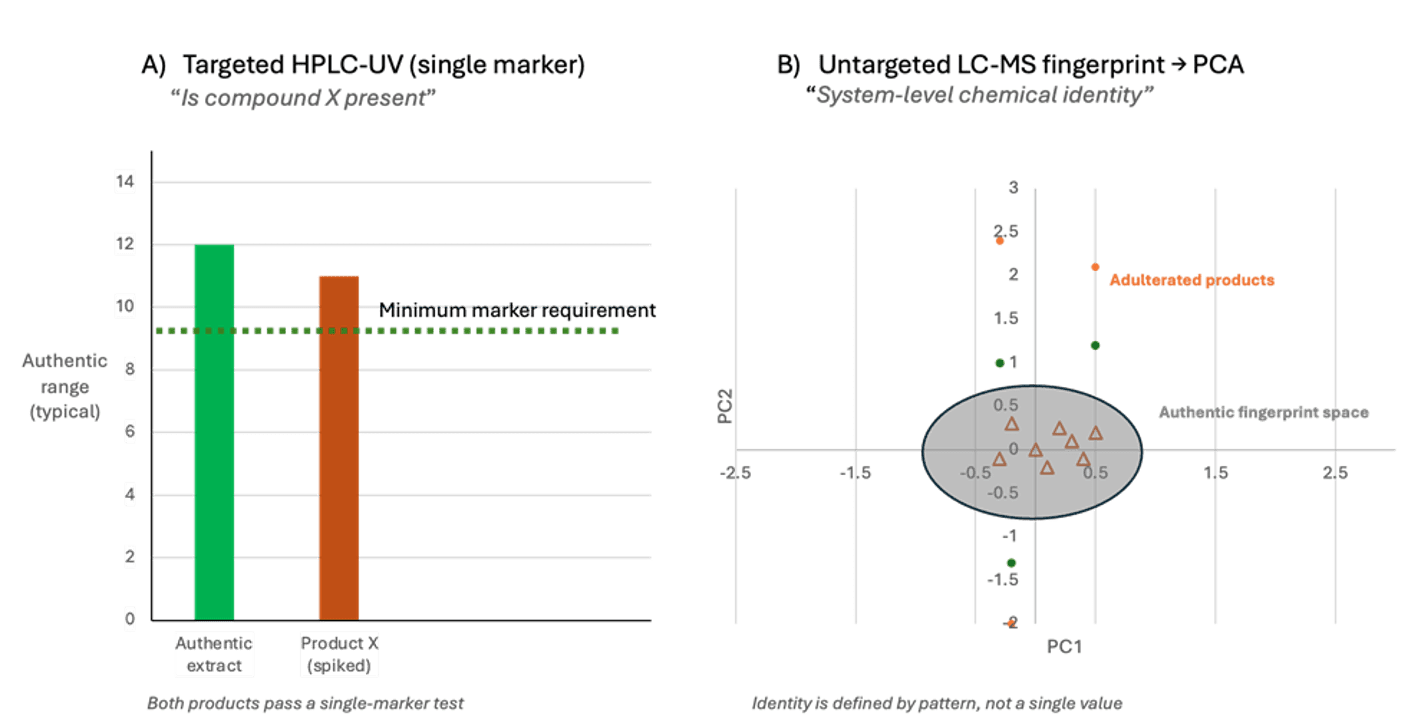
Figure 3 – From single markers to system-level identity. Left: A traditional single-marker test asks whether a target compound (e.g., compound X) falls within the expected concentration range. Both an authentic extract and a spiked product (Product X) pass, illustrating how this approach can fail to distinguish genuine material from engineered imitations. Right: A Principal Component Analysis (PCA) score plot based on untargeted metabolomic profiling reveals the full chemical picture. Authenticated botanical samples (triangles) cluster within a coherent fingerprint space defined by natural variation, while spiked products (circles) fall outside this region — even when they meet a single-marker specification. Identity is defined by pattern, not a single value.
Untargeted metabolomic profiling resolves this limitation by capturing the full chemical composition of the extract and interpreting it using multivariate statistical methods such as Principal Component Analysis (PCA). As shown in Figure 3b, genuinely authenticated botanical samples cluster within a coherent chemical space shaped by natural biological variation, while materials that have been diluted, substituted, or engineered to satisfy a narrow marker specification fall outside this region, even when they comply with single‑compound criteria. In this framework, identity is no longer defined by the presence or concentration of an isolated compound, but by whether the pattern of chemistry aligns with that of real plant material.
Overcoming Variability, Processing, and Adulteration
By shifting quality from single measurements to whole-pattern behaviour, metabolomics directly addresses the core problems that undermine botanical credibility: environmental and seasonal variability, processing effects, and adulteration. Earlier we saw how geography alters plant chemistry, how different plant parts and extraction methods reshape composition, and how products can be engineered to satisfy a narrow specification. Fingerprinting reframes these challenges. Instead of forcing complex biology into a fixed number, metabolomic models learn how authentic plants vary and define the natural “cloud” they occupy. This allows regional/environmental conditions to be differentiated as well as adulteration.
This principle is already playing out in practice. In Panax ginseng, LC–MS metabolomics showed that roots grown under different forest canopies formed distinct but overlapping clusters, capturing the chemical impact of environment while still separating genuine ginseng from non-ginseng material 20. In Saw Palmetto, 1H NMR fingerprinting grouped commercial products by extraction method and exposed samples that were diluted with cheaper vegetable oils, an artefact invisible to fatty-acid testing alone 22. In black cohosh, metabolomic profiles reliably distinguished Actaea racemosa from closely related Asian species in finished supplements, even where DNA barcoding failed due to processing 21.
Turmeric shows the same vulnerability: products can meet curcuminoid specifications while diverging from authentic Curcuma chemistry through the addition of dyes or foreign starches; untargeted LC–MS places these materials outside the natural chemical space defined by real rhizome extracts 23. Each case reflects a problem described earlier: geography, processing, substitution, and engineered compliance – each demonstrates how fingerprinting restores meaning to identity.
Regulation, Traceability, and Ethical Sourcing
Metabolomics sits awkwardly within most current regulatory frameworks, which remain grounded in fixed identities and single-parameter specifications. In the US, the FDA’s approach under DSHEA relies on declared species, GMP compliance, and targeted identity tests 24. In the UK, botanical products are assessed through MHRA and food standards pathways that similarly depend on pharmacopeial methods and marker compounds 25. EFSA’s evaluations across the EU are anchored to known constituents and safety thresholds. On the other hand regulators in South America, such as ANVISA in Brazil or CONAL in Argentina, operate through monographs, species declarations, and defined assays 23,26,27.
In the Asia-Pacific region, regulatory approaches are equally varied. Japan separates traditional medicines, regulated as drugs, from functional foods assessed under its FOSHU system 28. China’s State Administration for Market Regulation overseas both traditional Chinese medicine and health food registration 29, while India’s FSSAI governs botanical supplements as part of its broader food safety framework 30. Australia’s Therapeutic Goods Administration takes a comparatively stringent approach, requiring listed or registered product status for complementary medicines 31. Figure 4 shows the global regulatory landscape for botanical supplements. These systems are effective for ingredients that behave like chemicals. They are less equipped to accommodate evidence that expresses authenticity as a multivariate pattern rather than a single value.
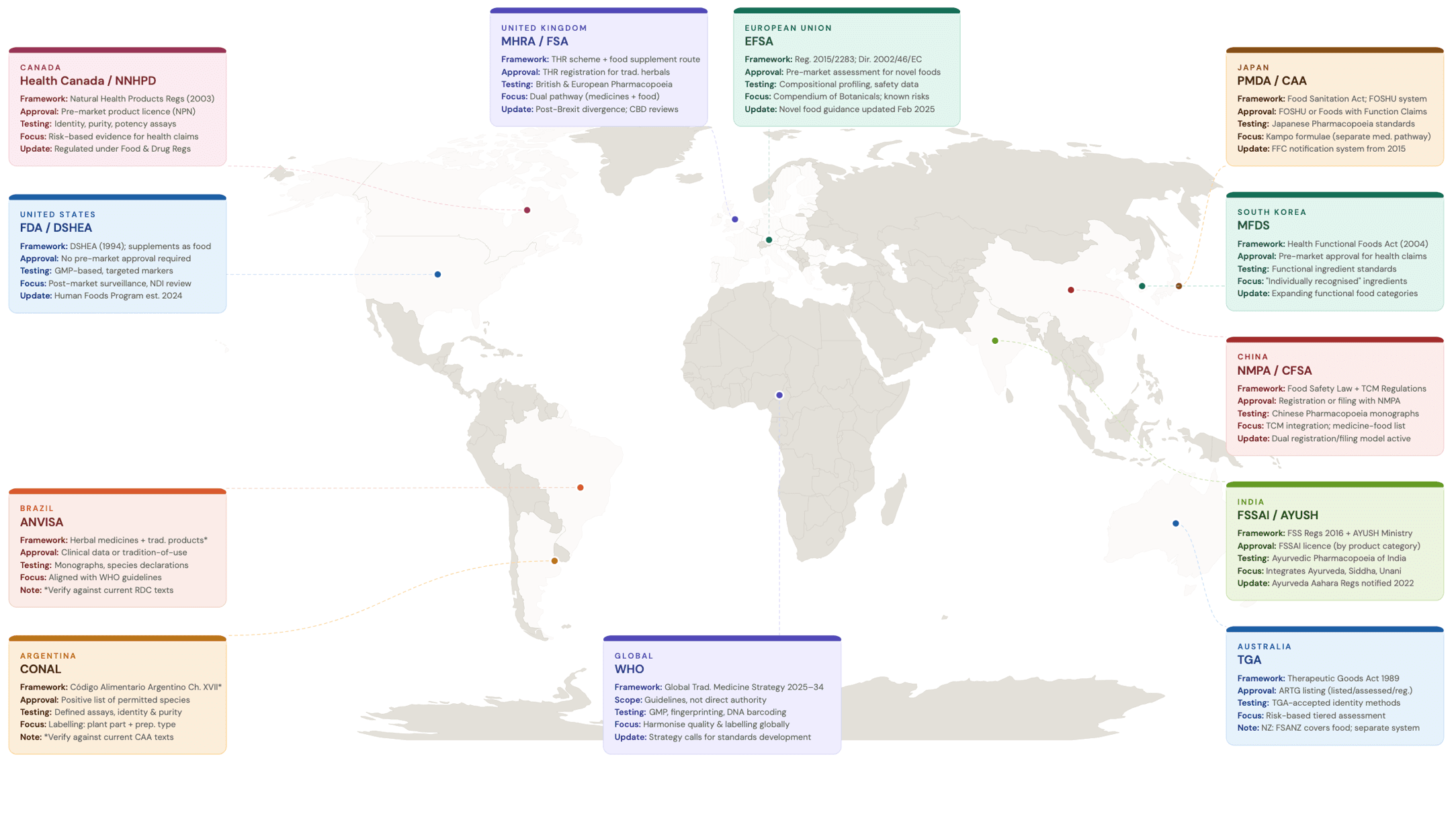
Figure 4 – Global regulatory landscape for botanical supplements. Compiled from national regulatory authority publications and comparative reviews 3,27,31–35.
Yet that very difference is what gives metabolomics its broader significance. When reference libraries are built from well-documented plant material, chemical fingerprints can link finished products back to biological and geographic origin, creating continuity across the supply chain. This capability aligns closely with the aims of the Nagoya Protocol, which seeks fair and equitable benefit-sharing from the use of genetic resources 36. By anchoring products to authentic plant populations and documented provenance, metabolomic frameworks can reinforce claims of origin, deter substitution, and support sourcing models in which value flows back to the communities and ecosystems from where these botanicals originate from 37.
Future Perspectives: From Verification to Design
Looking forward, metabolomics is poised to reshape how botanicals are verified, but also how they are developed and applied. As datasets expand and chemical fingerprints are increasingly linked to biological outcomes, it becomes possible to move beyond generic claims toward evidence-based designs, identifying which metabolic patterns correlate with cognitive resilience, immune modulation, or stress adaptation.
Emerging work already shows that subtle shifts across networks of metabolites, rather than single “actives,” underpin many of these effects, particularly in complex systems such as the gut–brain and immune axes 38. In this context, metabolomics becomes a bridge between traditional botanicals and modern biotechnology: enabling targeted cultivation, optimised processing, and, ultimately, more personalised nutrition strategies that align plant chemistry with individual physiology.
Contributor:
-
References
- Malekijahan F, Razavi SH, Nouri M, et al. (2025) Unlocking nature’s potential: The power of adaptogens in enhancing modern health and wellness. J Agric Food Res 24: 102501. DOI: 10.1016/j.jafr.2025.102501
- Colombo F, Restani P, Biella S, et al. (2020) and Regulatory Aspects. Appl Sci 10: 2387. DOI: 10.3390/app10072387
- Thakkar S, Anklam E, Xu A, et al. (2020) Regulatory landscape of dietary supplements and herbal medicines from a global perspective. Regul Toxicol Pharmacol 114: 104647. DOI: 10.1016/j.yrtph.2020.104647
- Straits research (2025) Botanical Supplements Market Size, Share & Growth Report by 2034. Last accessed April
- Virtue Market Research. (2024) Europe Herbal Supplements Market| Size, share, growth | 2024 – 2030. Last accessed April
- Market Data Forecast (2025) Latin America Herbal Supplements Market Size & Share, 2033. Last accessed April
- Surwase A. (2025) Herbal Supplements Market Size, Share & Growth Report, 2032.
- El-Saadony MT, Saad AM, Mohammed DM, et al. (2025) Medicinal plants: bioactive compounds, biological activities, combating multidrug-resistant microorganisms, and human health benefits – a comprehensive review. Front Immunol 16: 1491777. DOI: 10.3389/fimmu.2025.1491777
- Arumugam V, Vijayakumar V, Balakrishnan A, et al. (2024) Effects of Ashwagandha (Withania Somnifera) on stress and anxiety: A systematic review and meta-analysis. Explore 20: 103062. DOI: 10.1016/j.explore.2024.103062
- Wang Z, Singh A, Jones G, et al. (2021) Efficacy and Safety of Turmeric Extracts for the Treatment of Knee Osteoarthritis: a Systematic Review and Meta-analysis of Randomised Controlled Trials. Curr Rheumatol Rep 23: 11. DOI: 10.1007/s11926-020-00975-8
- Harnett J, Oakes K, Carè J, et al. (2020) The effects of Sambucus nigra berry on acute respiratory viral infections: A rapid review of clinical studies. Adv Integr Med 7: 240–6. DOI: 10.1016/j.aimed.2020.08.001
- Ichim MC. (2019) The DNA-Based Authentication of Commercial Herbal Products Reveals Their Globally Widespread Adulteration. Front Pharmacol 10: 1227. DOI: 10.3389/fphar.2019.01227
- Khan A, Kanwal F, Ullah S, et al. (2025) Plant Secondary Metabolites—Central Regulators Against Abiotic and Biotic Stresses. Metabolites 15: 276. DOI: 10.3390/metabo15040276
- Lan Y, Zhang M, Han M, et al. (2023) Differences in the Quality, Yield, and Soil Microecology of Ginseng in Different Planting Environments. Horticulturae 9: 520. DOI:10.3390/horticulturae9040520
- Kumar P, Banik SP, Goel A, et al. (2023) A critical assessment of the whole plant-based phytotherapeutics from Withania somnifera (L.) Dunal with respect to safety and efficacy vis-a-vis leaf or root extract-based formulation. Toxicol Mech Methods 33: 698-706. DOI: 10.1080/15376516.2023.2242933
- Ye L, Neilson A. (2016) Comparison of A-type Proanthocyanidins in Cranberry and Peanut Skin Extracts Using Matrix Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry. J Mol Genet Med 10: 1000209. DOI: 10.4172/1747-0862.1000209
- Phair J. (2023) Gas Chromatography-Mass Spectrometry (GC-MS): An Overview. Mass Spectrom Purif Tech. 9: 1000187. DOI:10.35248/2469-9861.23.9.187.
- Kumar BR. (2017) Application of HPLC and ESI-MS techniques in the analysis of phenolic acids and flavonoids from green leafy vegetables (GLVs). J Pharm Anal 7: 349–64. DOI: 10.1016/j.jpha.2017.06.005
- Dunn WB, Bailey NJC, Johnson HE. (2005) Measuring the metabolome: Current analytical technologies. Analyst 130(5): 606–625. DOI: 1039/B418288J
- Lee DY, Kim JK, Shrestha S, et al. (2013) Quality evaluation of Panax ginseng roots using LC‑QTOF/MS‑based metabolomics. Molecules 18: 14849–14861. DOI: 3390/molecules181214849
- Harnly J, Chen P, Sun J, et al. (2016) Comparison of flow‑injection MS, NMR, and DNA sequencing for authentication of black cohosh. Planta Med 82: 250–262. DOI: 1055/s-0035-1558113
- Booker A, Suter A, Krnjic A, et al. (2014) Phytochemical comparison of saw palmetto products using GC and ¹H‑NMR metabolomics. J Pharm Pharmacol 66: 811–822. DOI: 1111/jphp.12198
- Gafner S, Orhan N, Kahraman Ç, et al. (2025) A scoping review of turmeric adulteration across six continents. Pharm Biol 64: 87–107. DOI: 1080/13880209.2025.2606229
- Shipkowski KA, Betz JM, Birnbaum LS, et al. (2018) Naturally complex: Challenges in botanical dietary supplement safety assessment. Food Chem Toxicol 118: 963–971. DOI: org/10.1016/j.fct.2018.04.007
- Li S, Han Q, Qiao C, et al. (2008) Chemical markers for quality control of herbal medicines. Chin Med 3: 7. DOI: 1186/1749-8546-3-7
- European Food Safety Authority (EFSA). (2026) Botanicals. Available from: efsa.europa.eu. Last accessed April 28, 2026.
- Carvalho ACB, Ramalho LS, Marques RFO, et al. (2014) Regulation of herbal medicines in Brazil. J Ethnopharmacol 158: 503–506. DOI: 1016/j.jep.2014.08.019
- Blaze J. (2021) Comparison of regulatory frameworks for nutraceuticals in Australia, Canada, Japan and the United States. Innov Pharm 12: 1-8. DOI: 24926/iip.v12i2.3694
-
Ettinger DJ, Xin Li J. (2025) China: Master the ingredient regulations for health foods. Last accessed April 28, 2026.
- Food Safety and Standards Authority of India (FSSAI). (2016) Food Safety and Standards (Health Supplements, Nutraceuticals, Food for Special Dietary Use, Food for Special Medical Purpose, Functional Food and Novel Food) Regulations. New Delhi (India): Government of India. Last accessed April 28, 2026.
- Australian Government Department of Health. (2021) Therapeutic Goods Administration (TGA). Last accessed April 28, 2026.
- Low TY, Wong KO, Yap ALL, et al. (2017) Regulatory frameworks for risks associated with botanical food supplements. Compr Rev Food Sci Food Saf 16: 821–834. DOI: 1111/1541-4337.12289
- World Health Organization. (2024) Global traditional medicine strategy 2025–2034. Last accessed April 28, 2026.
- European Parliament and Council. (2002) Directive 2002/46/EC on food supplements. Last accessed April 28, 2026.
- Government of Canada Legislation Services Branch. (2025) Natural health products regulations. Last accessed April 28, 2026.
- Convention on Biological Diversity Secretariat. (2026) Nagoya Protocol on access and benefit‑sharing. Last accessed April 28, 2026.
- Alum EU, Manjula VS, Uti DE, et al. (2025) Metabolomics‑driven standardization of herbal medicine. Nat Prod Commun 20: 1934578X251367650. DOI: 1177/1934578X251367650
- Huang Y, Sun X, Huang Q, et al. (2024) Circulating metabolome in relation to cognitive impairment in older adults. Transl Psychiatry 14: 469. DOI: 1038/s41398-024-03147-9